Dea Biennial Inventory Form - Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from all other drugs or. The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each.
Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from all other drugs or. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients);
Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from all other drugs or. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location.
Dea Controlled Substance Log Template JMT Printable Calendar
Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. (2) schedule i and ii drugs must.
Dea form 41 Fill out & sign online DocHub
(2) schedule i and ii drugs must be separated from all other drugs or. Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. Modify eligible dea registration to.
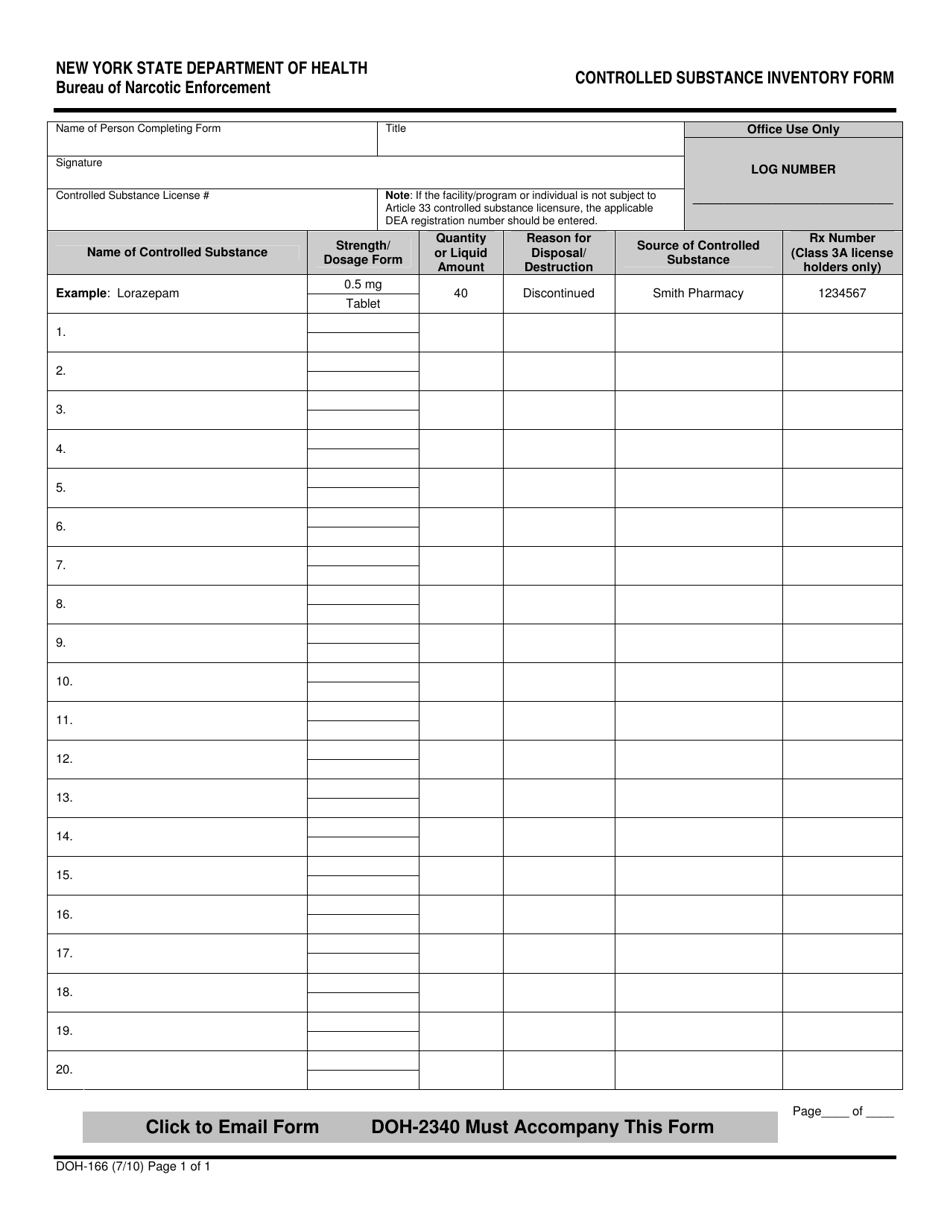
Form DOH166 Fill Out, Sign Online and Download Fillable PDF, New
(2) schedule i and ii drugs must be separated from all other drugs or. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. (2) schedule i and ii drugs must be separated from all other drugs or. Template (word) used to perform a biennial inventory of the.
SOLUTION Cs biennial inventory form Studypool
Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. (2) schedule i and ii.
CS Form E Biennial Inventory Environment, Health and Safety
(2) schedule i and ii drugs must be separated from all other drugs or. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. The dea requires a physical.
5 Best Printable Home Med Printablee 15048 Hot Sex Picture
The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from all other drugs or. Template (word) used to perform a biennial inventory of the controlled substances.
Dea biennial inventory form Fill out & sign online DocHub
Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from all other drugs or. Template (word) used to perform a biennial inventory of the.
DEA Biennial Controlled Substance Inventory Form Fill and Sign
Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); (2) schedule i and ii drugs must be separated from all other drugs or. (2) schedule i and ii drugs must be separated from.
Fillable Online Biennial & Initial Controlled Substance Inventory Form
(2) schedule i and ii drugs must be separated from all other drugs or. The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. Template (word) used to.
Fillable Online DEA Controlled Substance Inventory Form Fax Email Print
(2) schedule i and ii drugs must be separated from all other drugs or. The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Dea biennial controlled substance inventory form for the use of controlled substances in research a separate initial inventory is required for each. Modify eligible dea registration.
(2) Schedule I And Ii Drugs Must Be Separated From All Other Drugs Or.
Modify eligible dea registration to collect pharmaceutical controlled substances from ultimate users (e.g., patients); The dea requires a physical inventory of all controlled substances to be conducted every two years for each registered location. Template (word) used to perform a biennial inventory of the controlled substances currently stored at the dea registered location. (2) schedule i and ii drugs must be separated from all other drugs or.